Interactive Optical Science
Color Stability Science: The Physics of Pink Quartz Sun Fading
A vibrant pink specimen placed by a window slowly transforms into an opaque, ghostly white over several months. This is a fascinating demonstration of Photosensitive Gemstones reacting to ultraviolet light at an atomic level. Stop blaming "negative energy" for the loss of color; start examining the mineral lattice.
⚗️ Veteran Insight: The Photobleaching Reality Check ▼
I clearly recall the sinking feeling in 2018 when I lifted a prized Madagascan crystalline pink quartz from an east-facing display shelf. When I turned the specimen over, the contrast was violently apparent. The side facing the wall retained a deep, vitreous rose hue. The side facing the morning sun had bleached to the color of dirty snow. It felt chalky to the imagination, stripped of its visual depth. That single oversight taught me a harsh lesson about the unforgiving nature of photobleaching.
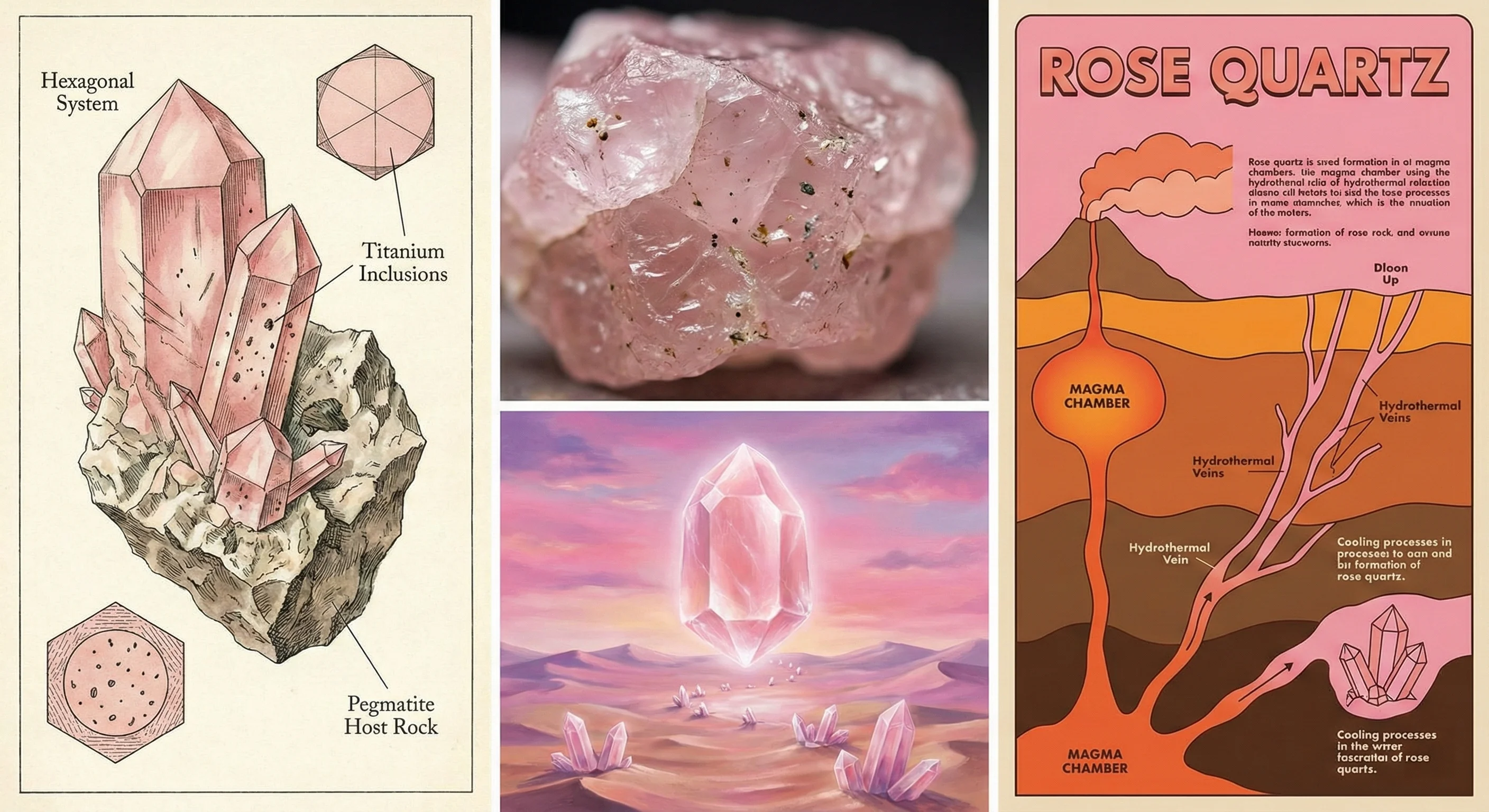
The ugly truth of quartz collecting is that we are constantly fighting entropy. We often see generalized advice suggesting that "keeping crystals out of direct sunlight" is sufficient. It is not. To understand Quartz Color Stability, you must understand the concept of the Radiation Color Center. In crystalline pink quartz, the color is not a physical pigment you can scrape off. It is an atomic anomaly.
Millions of years ago, natural gamma radiation from the surrounding earth crust irradiated the quartz lattice. This radiation possessed enough electron-volt energy to knock an electron out of a bonded oxygen atom near a phosphorus or aluminum impurity. This creates a "hole" — an unpaired electron state that absorbs specific wavelengths of visible light, reflecting back that delicate pink.
When you place this stone on a windowsill, you are bombarding it with high-energy UV photons. These photons excite electrons in the crystal, giving them enough kinetic energy to fall back into those "holes." The structural anomaly is neutralized. The color center is destroyed. The pink vanishes. This process is entirely irreversible through natural ambient means. You cannot "recharge" a bleached lattice by leaving it under the moonlight, despite popular options suggested in community forums. Moonlight simply lacks the ionizing radiation required to displace an electron. You trade temporary display aesthetics for the permanent destruction of geological history.